Innovative study testing AI for radiotherapy planning opens: the ARCHERY Study
November 10, 2022
A new study testing an innovative approach to radiotherapy treatment planning is opening: the ARCHERY study evaluates the use of artificial intelligence (AI) to automate several steps in the radiotherapy treatment pathway. It will assess both its quality, time and cost savings compared to the standard manual approach.
Radiotherapy is an effective treatment for many cancers. But planning a treatment that can deliver radiation to the tumour, while avoiding damage to healthy organs nearby, is a labour-intensive and highly skilled process. It requires input from expert clinicians and physicists or radiographers. This process is particularly challenging in low and middle-income countries, where there are not enough people with the necessary training and skills.
ARCHERY will assess the quality of radiation plans prepared using AI. It will also gather data on the time and cost involved, to allow evaluation of the health-economic impact of this approach.
If successful, the approach could transform radiotherapy treatment planning, speeding up the process, reducing costs and allowing more patients to access treatment. It could also help improve the consistency and quality of treatment as well as enable more complex techniques to be adopted.
The AI software being used iwithin ARCHERY has been developed by the MD Anderson Cancer Center. If found to be effective, it will be made available as a web-based not-for-profit service for public-sector and non-profit hospitals in low- and middle-income countries.
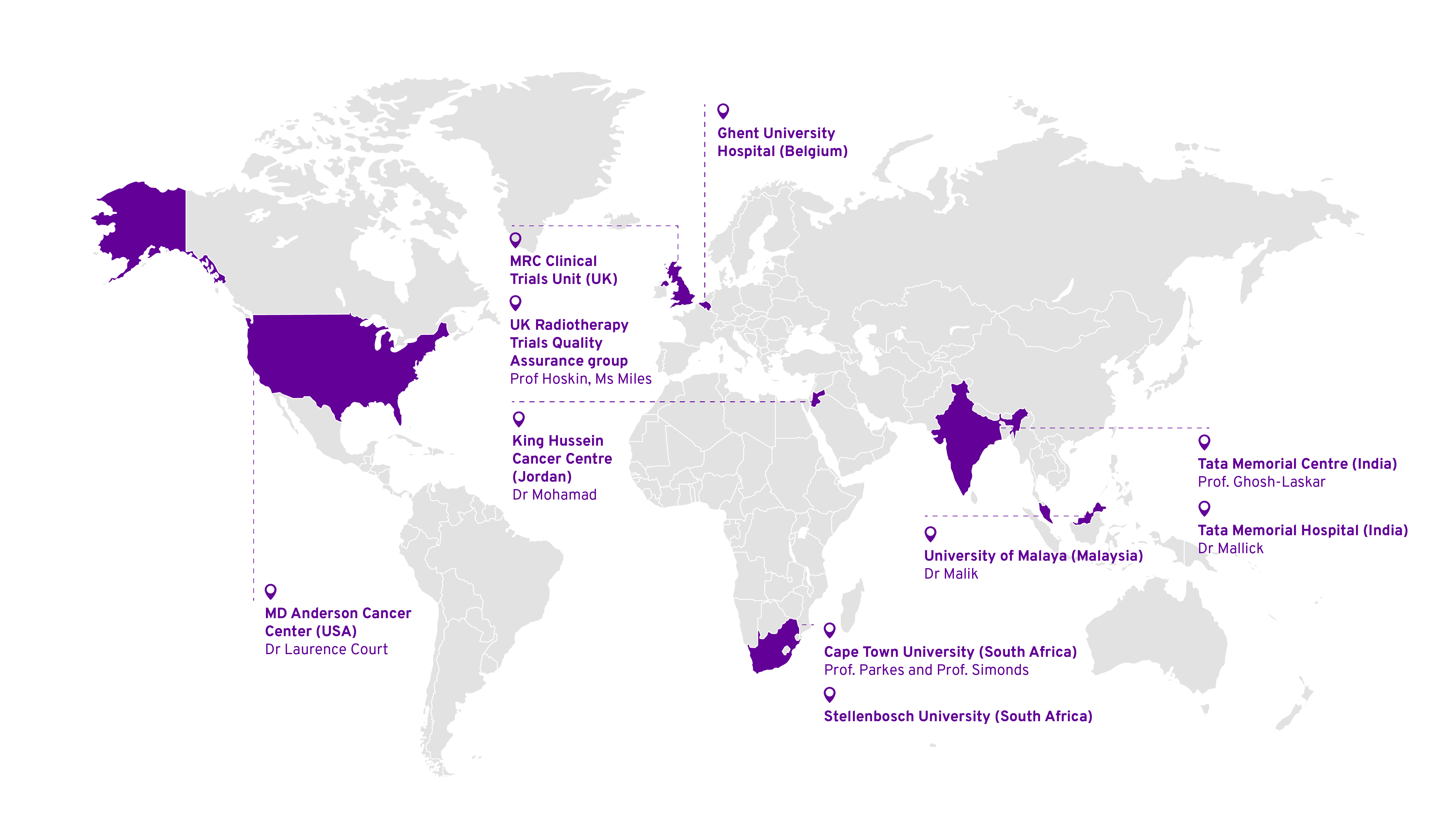
ARCHERY is the first study of its kind. It will look at treatments for three high burden tumours in a global setting: over 1000 patients with head and neck cancer, cervical cancer and prostate cancer will be enrolled in hospitals in India, South Africa, Jordan and Malaysia.
The ARCHERY study, led by Dr Ajay Aggarwal, Consultant Clinical Oncologist at Guy’s Cancer Centre/London School of Hygiene and Tropical Medicine and coordinated by the MRC Clinical Trials Unit at University College London, is funded by the US National Institute for Health and the Rising Tide Foundation for Clinical Cancer Research (RTFCCR).
RTFCCR supports the prostate cancer arm of the study. This high burden cancer is the most frequently diagnosed in men. In low-and-middle income countries radiotherapy is the major treatment modality for it, given the paucity of urological services and the consequent predominance of advanced cancers. In addition, poor quality radiotherapy planning is directly linked to an increased risk of cancer recurrence and functional morbidity for patients, such as bladder and bowel toxicity and sexual dysfunction. For these reasons, the ARCHERY study represents a great opportunity for enabling implementation of cheaper, easier, and more effective radiotherapy practice also for prostate cancer treatment in low- and medium-income countries.
Collaborating centres and partners
- Cape Town University (South Africa), Prof. Parkes
- Ghent University Hospital (Belgium), Prof Lievens
- King Hussein Cancer Centre (Jordan), Dr Mohamad
- MD Anderson Cancer Center (USA), Dr Court
- Stellenbosch University (South Africa), Prof, Simonds
- Tata Memorial Centre (India), Prof. Ghosh-Laskar
- Tata Memorial Hospital (India), Dr Mallick
- UK Radiotherapy Trials Quality Assurance group (RTTQA), Prof Hoskin, Ms Miles
- University of Malaya (Malaysia), Prof. Malik
RTFCCR
About the Rising Tide Foundation for Clinical Cancer Research
Rising Tide Foundation for Clinical Cancer Research (RTFCCR) is a charitable, non-profit organization established in 2010 and located in Schaffhausen Switzerland. RTFCCR’s primary consideration in granting support is given to truly innovative, unique, patient-centered clinical research. The long-term ambition of the foundation is to optimize partnerships and attract the best in Phase I to Phase III clinical trials that aim to bring maximum patient benefit in the shortest time possible. With patients at the core of the mission, the foundation strives to support clinical trials resulting in the creation of less toxic therapeutic approaches, better disease burden management, earlier cancer detection, and innovative intervention strategies that will lead to increased quality of life and survival.