Science of Early Intervention
Within the Science of Early Intervention Focus Area, RTFCCR intends to support interventional clinical trials and clinical validation studies exploring novel approaches to act on precursors or early cancer stages, interfering with their development towards more aggressive cancer stages.
Applicants with questions about the suitability of their project are welcome to contact us at rtfccrteam@risingtide.ch.
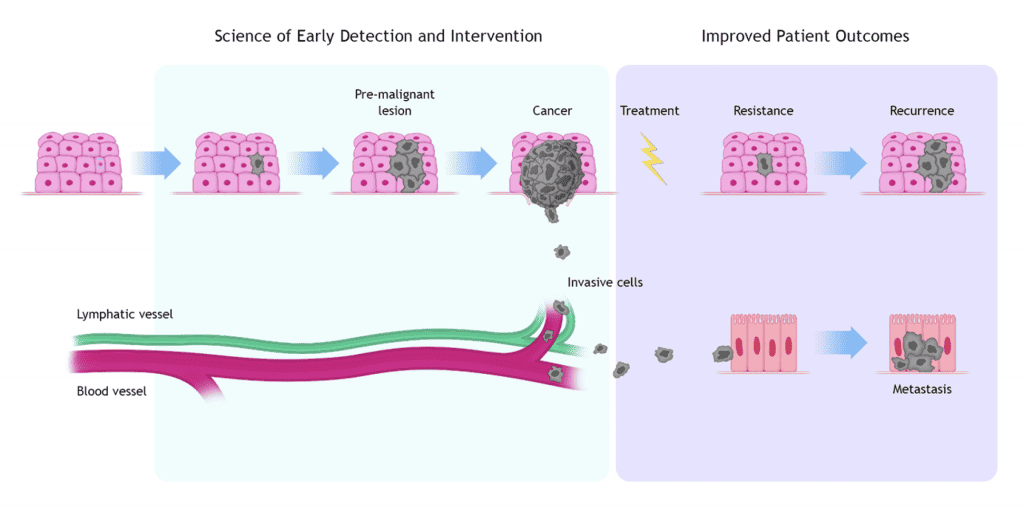
Stages of the Cancer Journey
- A tumor originates within normal tissues when one or more cells experience aberrant genetic events and start proliferating in an uncontrolled manner.
- This change in cell behavior is caused by a genetic event (a mutation). The cause of the cancer-causing mutation can be sporadic or due to environmental factors (carcinogens, UV, etc.)
- These abnormally growing cells (dysplasia) can develop into a mass (in situ) that can start interacting with the surrounding cells (tissue microenvironment).
- The in-situ tumor can further develop, becoming an invasive tumor. Through cell signaling, the tumor stimulates capillary growth to have access to nutrients in the blood. Moreover, it also convinces the body’s immune system (lymphocytes and macrophages) to support its growth instead of fighting it.
- When the change in cell behavior reaches a certain stage, it can be detected, and external interventions can be used to eradicate it.
- In some cases, the intervention (e.g., neo-adjuvant, chemotherapy, radiotherapy, surgery, adjuvant treatments) does not eradicate the tumor: some cells can be left behind and, with time, begin proliferating again, resulting in tumor recurrence or relapse.
- Sometimes before, during or after treatment, the tumor can shed living cells in blood or lymphatic vessels, which can move to colonize other tissues (metastasis)
The Rising Tide Foundation for Clinical Cancer Research (RTFCCR) seeks to support clinical cancer research across different stages of the cancer journey, with specific focus on the following areas:
- Science of Early Detection and Intervention, where support is provided for studies aimed at detecting abnormal cells at the earliest time possible and intervening to eliminate them before they can develop into more aggressive tumor stages.
- Improved Patient Outcomes, where support is provided for interventional clinical trials that advance therapeutic approaches with the best possible outcomes and the least toxicity.
Funding Opportunities
Open Call
We accept Letters of Intent (LOIs) all year round with cutoff deadlines for internal assessment three times per year. Once you have submitted your LOI, you will receive further information regarding timing and next steps.
Pre-application Grants
Pre-application grants provide gap funding to support patient experts’ helping to develop grant applications/ protocols.